Dichloroacetate
Drug Summary: Dichloroacetate stimulates an enzyme called PDC that is essential for the production of energy in cells. Because inefficient energy production is believed to contribute to the progression of HD, dichloroacetate therapy could result in increased energy production, and could possibly help delay HD progression.
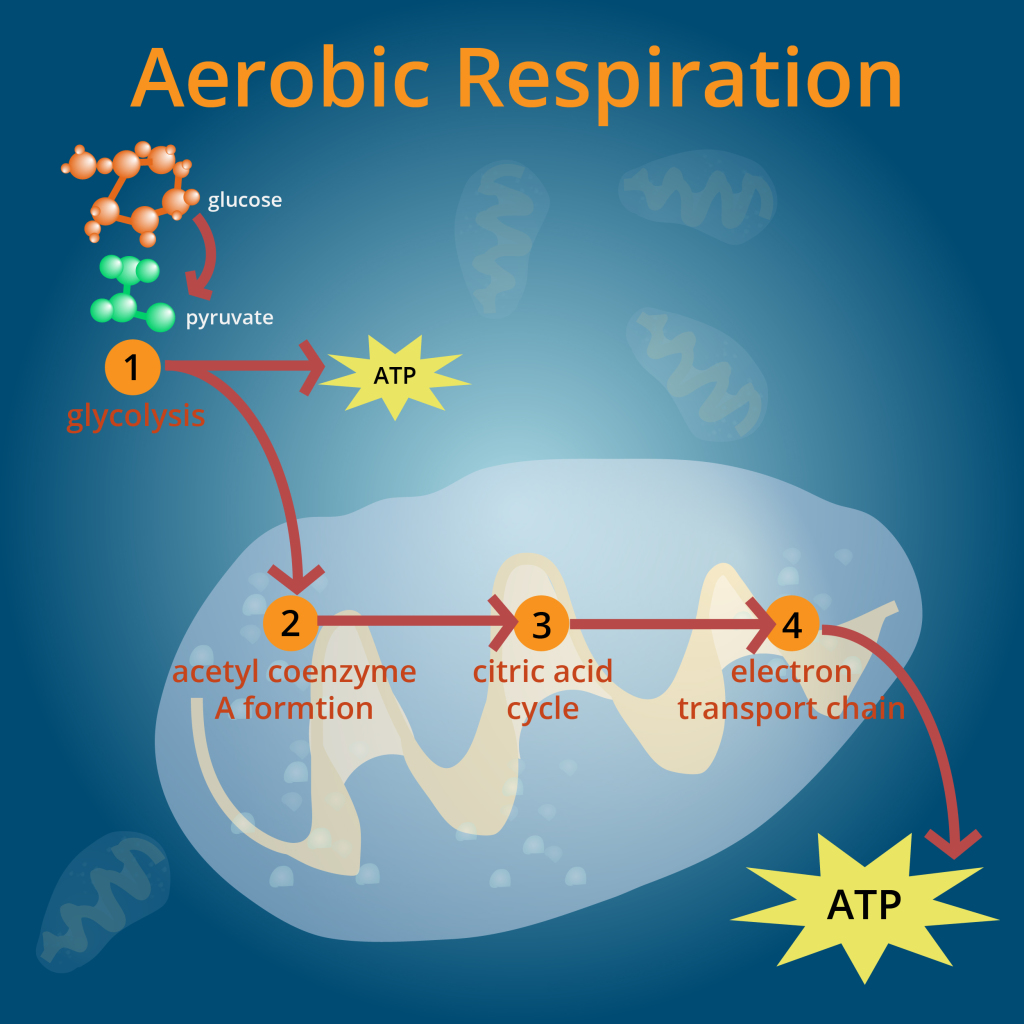
The altered huntingtin protein seen in the nerve cells of people with HD has been known to cause a decrease in the amount of energy available in cells by disrupting energy metabolism. (For more on metabolism, click here.) The mitochondria of HD cells appear to be damaged by the altered huntingtin and are unable to perform aerobic respiration, a form of energy metabolism. The mitochondrial damage forces cells to resort to anaerobic respiration, a less efficient form of energy metabolism. The inability to perform efficient aerobic respiration leads to decreased energy production. This energy deficit in HD cells leads to various consequences: the cell is unable to perform its different functions as efficiently as it used to and is more vulnerable to toxicity by various molecules.
Researchers believe that increasing the efficiency of aerobic respiration, and in turn, increasing the energy available to the cell, is one way of slowing the progression of HD.
One way by which scientists measure the efficiency of metabolism is cells is by measuring the cells’ lactate levels. Lactate, a by-product of anaerobic respiration, is often found in higher concentrations in cells with decreased metabolism efficiency. High levels of lactate indicate that anaerobic respiration (the less efficient form of energy production) is the primary form of metabolism. On the other hand, low lactate levels indicate that aerobic respiration is the primary form of metabolism used by the cells.
Dichloroacetate in energy metabolism^
Dichloroacetate has been found to decrease lactate production in cells by stimulating the pyruvate dehydrogenase complex (PDC), a critical group of enzymes involved in energy metabolism. The PDC is a large complex that is composed of multiple copies of three enzymes – E1, E2, and E3. The PDC serves as the vital enzyme involved in pyruvate oxidation, the step in aerobic respiration in which pyruvate is converted to acetyl-CoA. Pyruvate is a product of glycolysis, the first step in energy metabolism where sugar molecules from the carbohydrates we eat are transformed into pyruvate to be used for further processing in metabolism.
Each of the three enzymes that make up the PDC performs specific reactions that collectively transform pyruvate to acetyl-CoA. Acetyl-CoA is then transported into the mitochondria and enters the Kreb’s Cycle, a step in aerobic respiration. Once acetyl-CoA enters the Kreb’s Cycle, it undergoes various reactions that ultimately end in the production of large quantities of ATP. The PDC acts as a gatekeeper that facilitates and regulates the entry of pyruvate in to the Kreb’s Cycle.
In essence, the PDC determines whether the pyruvate molecules will be transformed into acetyl-CoA. If pyruvate is converted to acetyl-CoA, the cells can use the acetyl-CoA to undergo aerobic respiration. If pyruvate is unable to be converted to acetyl-CoA, the pyruvate is used in anaerobic respiration. If the PDC is damaged, fewer pyruvate molecules are converted to acetyl-CoA, which results in a decrease in the rate of aerobic respiration and a decrease in the number of ATP molecules produced. Instead, the pyruvate molecules stay in the cytosol and undergo anaerobic respiration, producing increased amounts of lactate. An abnormal lactate buildup results in various symptoms such as severe lethargy (tiredness) and poor feeding, especially during times of illness, stress, or high carbohydrate intake.
How is PDC activity regulated?^
A family of enzymes called PDC Kinases acts to add phosphate groups to the E1 enzyme of the PDC. Adding a phosphate group to E1 inhibits the activity of the PDC complex. Acetyl-CoA usually activates these PDC kinases as a way to stop production of more acetyl-CoA when it is already present in large amounts and continued production is no longer needed.
Dichloroacetate therapy has been used to increase the efficiency of aerobic respiration. Researchers have reported that dichloroacetate stimulates the PDC by inhibiting the kinase that inactivates the PDC. Once the kinase is inhibited, the PDC continues to be activated and is able to perform its function of converting pyruvate to acetyl-CoA for use in aerobic respiration.
Given that impaired energy metabolism is implicated in the progression of HD, dichloroacetate treatment may improve metabolism and slow HD progression. In mouse models of HD, it is thought that the altered huntingtin protein interferes with the PDC kinases, causing a decrease in active PDC in nerve cells. This additional finding of decreased active PDC in HD nerve cells further supports the possibility of using dichloroacetate to stimulate the PDC and improve cell metabolism.
Dichloroacetate safety^
There is some concern about the toxicity of dichloroacetate. Accumulations of dichloroacetate in groundwater have been described by some reports as a potential health hazard. However, concern about dichloroacetate toxicity is mainly based on data obtained in rats who were administered dichloroacetate at doses thousands of times higher than those to which humans are usually exposed. In these animals, chronic administration of dichloroacetate was found to cause liver problems and tumors. (Stacpoole, 1998.) In contrast, the dosage given to most humans is much lower than that administered to the rats. In clinical trials where dichloroacetate is used as a medical drug, no major side effects have been reported. Dichloroacetate is currently the most effective treatment for a disease known as congenital lactic acidosis (CLA). People with CLA have defective PDC enzymes and are thus unable to efficiently produce energy. In one study, patients with CLA were treated with 25-50 mg of dichloroacetate per 1 kg of body weight. No major complications were observed in the participants. (Stacpoole, 1997.) However, more research is currently being done to study the possible toxicity of dichlororacetate.
Issues of dichloroacetate toxicity have also arisen in research not directly related to HD. Dichloroacetate has also been found to protect against neuronal damage in the striatum of rats whose nerve cells have been deprived of blood flow. (Peeling, et al., 1996.) However, a recent report on an ongoing trial of dicholoroacetate treatment in people with mitochondrial disorders has reported that some patients developed new pathological symptoms and some had worsening in the transmission of nerve impulses. (Haas, et al., 2000.) Long-term trials are necessary to clarify the side effects associated with dichloroacetate and its role in HD treatment.
Research on Dichlororacetate^
Gansted, et al. (1999) investigated whether dichloroacetate can improve the condition of people with mitochondrial myopathies (MM). The researchers hypothesized that dichloroacetate treatment in people with MM will result in improved energy metabolism. Because a decrease in metabolism is hypothesized to also be associated with HD, results of studies on MM and dichloroacetate may lead clues to the efficacy of dichloroacetate in HD treatment.
The mitochondrial myopathies are a group of neuromuscular diseases caused by damage to the mitochondria. Some of the more common mitochondrial myopathies include Kearns-Sayre syndrome, myoclonus epilepsy with ragged-red fibers (MERRF), and mitochondrial encephalomyopathy lactic acidosis and stroke-like episodes (MELAS). Mitochondrial myopathies are often caused by mutations in the DNA encoding the electron transport protein complexes, resulting in decreased ATP production. Aerobic respiration is not as efficient, so the cells of people with MM have to resort to more anaerobic respiration for their energy needs. The increased anaerobic respiration results in accumulations of lactate during exercise and contributes to exercise intolerance.
Dichloroacetate treatment was administered for 15 days to 7 people with MM. The study showed that dichloroacetate administration lowered lactate levels in most of the patients, indicating that dichloroacetate may improve metabolism efficiency. However, three patients reported that dichloroacetate caused a considerable sedative effect.
Andreassen, et al. (2001) reported that dichloroacetate has therapeutic effects in two mouse models of HD. One model, called the R6/2 mice, had C-A-G repeat lengths of 141 to 152. These mice exhibited HD-like symptoms such as decreased weight, motor dysfunction, brain atrophy, neuronal inclusions, and an increased occurrence of diabetes. The second mouse model, called the N171-82Q mice, had 82 C-A-G repeats in their Huntington genes. These mice exhibited symptoms similar to those of the R6/2 mice except that their symptoms were less severe and more delayed in onset.
Dichloroacetate treatment began at 4 weeks of age and was terminated at 12 weeks of age. A dose of 100mg/kg of body weight was administered daily. The study showed that dicholoroacetate-treated mice of both models showed significantly improved survival and motor function, as well as delayed weight loss and nerve cell loss. The development of diabetes was also delayed. Dichloroacetate was also found to maintain normal amounts of the active form of PDC. However, formation of neuronal inclusions was not altered by dichloroacetate treatment. The results of this study raise the possibility that dichloroacetate might be a potential HD treatment with therapeutic benefits for people with HD.
For further reading^
- Peeling, et al. Protective effect of dichloroacetate in a rat model of forebrain ischemia. Neuroscience Letters. 1996; 208: 21-24.
Peeling, et al. reported that dichloroacetate was able to protect against neuronal damage in the striatum of rats whose nerve cells have been deprived of blood flow. - Haas, et al. Results of the UCSD open label dichloroacetate trial in congenital lactic acidosis. In: Zullo SJ, ed. Mitochondrial Interest Group Minisymposium (Mitochondria: Interaction of Two Genomes). Bethesda, MD: NIH, 2000 p.2.
Haas, et al. reported that some patients treated with dichloroacetate had developed new pathological symptoms and some had worsening in the transmission of nerve impulses. - Gansted, et al. Dichloroacetate treatment of mitochondrial myopathy patients. Neurology. 1999; 52 (Suppl 2): A544.
This article reports that dichloroacetate treatment resulted in lowered lactate levels (and consequently, increased energy production) in people with mitochondrial myopathies. - Andreassen, et al. Dichloroacetate exerts therapeutic benefits in transgenic mouse models of Huntington’s disease. Annals of Neurology. 2001; 50(1): 112-6.
This article reports that dichloroacetate treatment resulted in various beneficial effects in mouse models of HD. - Stacpoole, et al. Clinical Pharmacology and Toxicology of Dichloroacetate. Environmental Health Perspectives. 1998; 106: Supplement 4.
This article reports that rats treated with dichlororacetate at dosages thousands of times higher than normally prescribed to humans exhibited various pathological side effects. - Stacpoole, et al. Treatment of congenital lactic acidosis with dichloroacetate. Archives of Disease in Childhood. 1997; 77: 535-541.
This article reports that dichloroacetate treatment resulted in lower lactate levels in people with congenital lactic acidosis.
-P. Chang, 7/5/04