Dimebon (Latrepirdine)
Update: According to a press release on 4/11/11, dimebon did NOT pass the Horizon trial and is not effective in treating HD. There was no statistically significant difference in symptoms between the experimental (received dimebon) and placebo (did not receive dimebon) groups. According to the president and chief executive officer of Medivation, development of dimebon in HD will be discontinued. However, dimebon trials for treatment of Alzheimer’s disease will continue.
The press release can be viewed here.
Drug Summary: Dimebon (pronounced deh-mah-bonn) is an anti-histamine drug with a 20-year history of use in Russia. Its potential as a neuroprotective agent was recently recognized when a large chemical library screen found that the class of molecules it belongs to, gamma-carbolines was likely to be effective in blocking nerve-cell receptors for glutamate (see Glutamate excitotoxicity below) and in blocking cholinesterases (see Low levels of Acetylcholine below). Studies are now being undertaken to assess its efficacy.
Currently, Dimebon is being developed by pharmaceutical company Medivation, in collaboration with Pfizer, for use in treating Huntington’s Disease (HD) and Alzheimer’s Disease. Lab studies (using rats and human nerve cells in culture) have shown that Dimebon may be helpful in treating problems common to the two diseases, including glutamate excitotoxicity and low levels of acetylcholine. Clinical studies with both Huntington’s and Alzheimer’s patients have also had encouraging results. Dimebon’s protective effects in these cases may make a significant difference in alleviating cognitive symptoms and improving overall quality of life. Dimebon is currently in Phase III clinical trials for both diseases.
Glutamate excitotoxicity
The chemical glutamate is an excitatory neurotransmitter in the brain, and a routine part of nerve impulse transmission. When present in excessive amounts though, glutamate can cause a chain of reactions that ultimately leads to nerve cell death. This process is called “excitotoxicity” because glutamate over-excites the nerve cell and becomes bad for it, or toxic. Cell death can also occur at normal glutamate levels if nerve cells become “hypersensitive” to glutamate. This hypersensitivity is thought to be the reason glutamate excitotoxicity exists in HD. For a more detailed explanation of this process, please see Glutamate Toxicity: Disease Mechanism V.
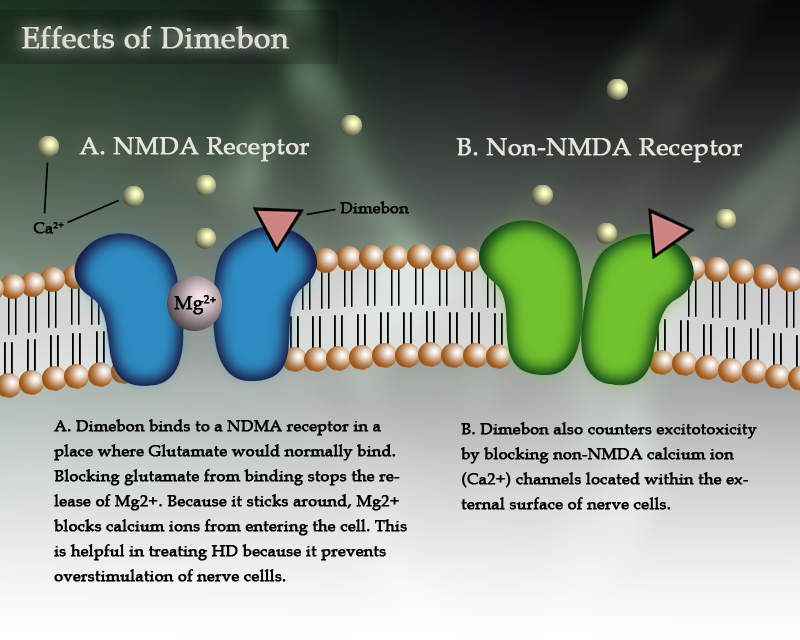
In rat studies, Dimebon has been shown to work against glutamate excitotoxicity in two main ways. First, Dimebon acts as an antagonist to glutamate by binding to glutamate receptors, called NMDA receptors, on the surface of nerve cells. If glutamate can’t bind to the NMDA receptors because Dimebon is in the way, then glutamate cannot send its message to the cell. This process is similar to the situation that would arise if you needed to send an e-mail, but someone was sitting in your chair. If the other person sits there and doesn’t send a message, then you can’t sit down to send a message and no message gets sent at all. This activity has the potential to prevent cell death if there are abnormally high levels of glutamate in the brain but also, as is most likely in Huntington’s, if nerve cells are hypersensitive to normal levels of glutamate.
Dimebon also counters excitotoxicity by blocking calcium ion (Ca2+) channels located on the surface of nerve cells. This activity prevents the influx of Ca2+ ions, a consequence of glutamate excitotoxicity. (For more information about this process please see Glutamate Toxicity: Disease Mechanism V.) When blocked, Ca2+ cannot get into the cell to activate free radicals and other damaging molecules that ultimately lead to cell death. In this way, Dimebon may be able to stop a cell from progressing towards cell death even if glutamate is able to over-excite it.
So Dimebon may help prevent nerve cell death by preventing glutamate from over-stimulating nerve cells and preventing over-excited cells from taking in large amounts of Ca2+. How else might Dimebon help treat HD?
Low levels of Acetylcholine
Acetylcholine is a neurotransmitter that is vital to proper memory function. Current research suggests that, in HD, degeneration of the basal ganglia involves the death of a great number of cholinergic nerve cells, which are the nerve cells that produce acetylcholine. This loss of cholinergic nerve cells results in low levels of acetylcholine, which likely contribute to the cognitive impairment associated with HD. Replacing acetylcholine by way of dietary supplements is sometimes suggested as a treatment for memory deficiency in neurodegenerative diseases.
Studies have suggested two ways that Dimebon might enhance cognition (memory and learning ability) in HD patients. The first of these is that Dimebon’s has been shown to protect nerve cells in culture from neurotoxins. The simple prevention of nerve cell death is surely beneficial to memory. Additionally, a high number of surviving cells can produce close to normal levels of acetylcholine. However, even if acetylcholine is only produced in small amounts, Dimebon can keep this neurotransmitter at normal levels by inhibiting two enzymes that normally break it down. This activity by Dimebon helps keep each molecule of acetylcholine around longer, allowing small amounts to accumulate and reach normal levels.
So we know that Dimebon can decrease cell death from excitotoxicity and that it can increase levels of acetylcholine. The question then becomes: how well might Dimebon work as a treatment for HD? The three studies outlined below—one on rats and two in humans—test Dimebon’s effect on the entire organism. These studies were designed and evaluated with Alzheimer’s disease in mind, but the results are still useful to HD research.
1) In a 2001 study by Bachurin, S. et al. (see “For further reading” below), rats were given a neurotoxin that selectively kills cholinergic nerve cells. Then one group of rats was treated with Dimebon and a second group was not. The group of rats that was given no treatment had severely reduced cognition. In comparison, rats that were treated with Dimebon showed significantly better memory and learning ability.
2) The second study was a preliminary clinical trial with 14 participants exhibiting mild to moderate Alzheimer’s disease. The study lasted 8 weeks but participants showed marked improvements in cognition and independence as early as 2-4 weeks. A variety of common conditions were alleviated including depression, anxiety, tearfulness, headache and psychopathic symptoms. These improvements tended to be gradual but significant; for example, 50% of patients with headaches showed improvement at 4 weeks and 80% showed improvement at 8 weeks. This trial was also part of the Bachurin, S. et al 2001 study mentioned above.
3) In September 2006, results of a phase II study of Dimebon were reported by a company called Medivation. According to standard procedures, phase II testing uses a medium-sized group of patients to test a drug’s usefulness and look for side effects. In this study, 183 individuals with mild to moderate Alzheimer’s disease were treated with either Dimebon or a placebo for six months. Five different tests were used to evaluate progression of the disease and Dimebon-treated participants showed “highly statistically significant improvement” on all of them. This means that it is very likely that patients treated with Dimebon improved because of the drug and not because of a placebo effect. Patients taking Dimebon not only did better than placebo patients throughout the trial but also actually improved from their original, or baseline, condition.
Dimebon was also well tolerated – more serious adverse events occurred in the placebo group. This is certainly an advantage of using a drug with a previous history of use in humans. A large percentage of individuals (86%) chose to participate in a continuation study that brought treatment to a full year. Results from this study were announced in June 2007 and demonstrated that at one year, the benefits of Dimebon over placebo on all five efficacy endpoints were stable or greater when compared to the benefits at six months.
Medivation is currently undertaking The Connection Study, a Phase 3 Clinical Trial of Dimebon in patients with mild to moderate Alzheimer’s Disease, and The Concert Study, a similar trial in Alzheimer’s patients who are also taking Aricept (donepezil).
So what about Huntington’s?
In July 2008, Medivation published the results of their Phase II trial of Dimebon in persons with HD. The trial was done in cooperation with the Huntington Study Group and enrolled 90 participants with mild to moderate HD for three months. The results showed statistically significant improvement in cognition as measured by the Mini-Mental State Examination (MMSE). This is a very exciting result because there is currently no treatment that has been shown to improve cognition in HD. Additionally, Dimebon was well-tolerated, showing very few side effects.
In July 2009, Medivation, teamed with a larger pharmaceutical company called Pfizer, announced their Phase III clinical trial, Horizon. Horizon will enroll 350 HD patients suffering from cognitive impairment, and will last for six months. As a Phase III trial, Horizon will focus on trying to determine whether or not Dimebon significantly improves cognition in HD patients and can be approved to treat cognitive symptoms of HD, such as memory and decision making.
You can stay updated on the progress of both Alzheimer’s and Huntington’s trials by checking press releases on the Medivation website or by returning to the HOPES site in the future when new results are published. We’ll do our best to keep you posted!
For further reading
- Bachurin S., Bukatina, E., Lermontova, N., Tkachenko, S., Afanasiev, A., Grigoriev, V., Grigorieva, I., Ivanov, YU., Sablin, S. & Zefirov, N.: Antihistamine Agent Dimebon As a Novel Neuroprotector and a Cognition Enhancer. Annals of the New York Academy of Sciences 939: 425-435, 2001
- This scientific study includes information on many aspects of Dimebon’s potential use in Alzheimer’s disease. It is easy to get bogged down in details and scientific language but if you stick to the introduction, results, and discussion you will find it informative.
- This is an easy-to-read press release by the company that owns rights to develop and test Dimebon as a neuroprotective drug in the US.
- Another easy-to-read press release from Medivation announcing the beginning of the Dimebon trial for Huntington’s Disease.
- Medivation (June 11, 2007). Medivation’s Dimebon(TM) Maintains Statistically Significant Benefit on All Five Efficacy Endpoints in Alzheimer’s Disease Trial After One Year of Therapy. Press Release.
- Click here to read this article.
- The Medivation press release announcing results of the year-long Dimebon trial for Alzheimer’s.
- Medivation (July 7, 2008) Medivation Announces Positive Top-Line Results From Phase 2 Dimebon Study in Huntington’s Disease. Press Release. Click here to read this article.
- The Medivation press release announcing results of the Phase II trial in Huntington’s Disease (HD)
-F. Clum, 4-19-10
-updated A. Zhang, 4-25-11
