Signaling through CD19 Activates Vav/Mitogen-Activated Protein Kinase Pathway and Induces Formation of a CD19/Vav/Phosphatidylinositol 3-Kinase Complex in Human B Cell Precursors
*
Wen-Kai Weng, Lisa Jarvis and Tucker W. LeBien
‡
From the Department of Laboratory Medicine and Pathology,
University of Minnesota Medical School,
Minneapolis, MN 55455
Journal of Biological Chemistry 269:32514 (1994)
ABSTRACT
The B cell-specific cell surface molecule CD19 plays a role in regulating immunoglobulin (Ig) receptor signaling, and cross-linking CD19 activates several signaling molecules in mature human B cells. In surface Ig negative B cell precursors, a protein tyrosine kinase (PTK)-dependent homotypic aggregation response can be triggered by cross-linking CD19. In the current study, we examined the outcome of PTK-mediated signal transduction following CD19 cross-linking on surface Ig negative and surface Ig positive B cell lines, as well as freshly isolated surface Ig negative B cell precursors. PTK activation resulted in the tyrosine phosphorylation of multiple protein substrates, and peaked at 0.5 to 1 min following CD19 cross-linking in all B-lineage cells examined. One of the tyrosine phosphorylated substrates was identified as the hematopoietic-specific protein Vav, a guanine nucleotide exchange factor that activates the Ras pathway. Evidence consistent with Ras pathway activation was also demonstrated by MEK activation and subsequent phosphorylation of a MAP kinase fusion protein. CD19 cross-linking, sequential immunoprecipitation, and Western blotting revealed that: a) Vav becomes associated with CD19, b) phosphatidylinositol 3-kinase (PI 3-kinase) becomes associated with CD19, and c) PI 3-kinase becomes associated with Vav. No such physical interaction occurred following control IgG1 cross-linking or cross-linking of class I major histocompatability complex cell surface molecules. Coupled with a previous report (Tuveson et al., Science 260:986, 1993), our data supports a model in which CD19 cross-linking induces the formation of a signaling complex that leads to the activation of two pathways involving Ras and PI 3-kinase.
INTRODUCTION
Analysis of the structure and function of cell surface molecules on human B lymphocytes continues to be an area of active investigation. One cell surface molecule that has been extensively studied is CD19. CD19 is expressed from early B cell precursors to mature B cells, and is lost at the terminal plasma cell stage (1,2). The human CD19 gene is composed of 15 exons and encodes a 540 amino-acid type I integral membrane glycoprotein (3). CD19 has an extracellular region which contains two immunoglobulin (Ig)
1-like domains, one transmembrane domain, and a 242 amino-acid cytoplasmic domain (3). The conserved amino acid sequence and length of the CD19 cytoplasmic domain between human, guinea pig, and mouse suggests that it plays a critical role in the transduction of CD19-mediated signals (4). CD19 contains nine cytoplasmic Tyr residues that can be potentially phosphorylated by protein tyrosine kinases (PTK), and some of these tyrosine-containing motifs may directly bind to SH2 domains in other signaling molecules (5,6). Indeed, phosphatidylinositol 3-kinase (PI 3-kinase) has been shown to directly bind to CD19 via two motifs with the amino acid sequence Tyr-X-X-Met (7).CD19 has been shown to be part of a multimeric protein complex on the cell surface of mature B cells, which includes complement receptor 2 (CD21), TAPA-1 (CD81) and Leu-13 (8,9). A model has been proposed whereby the CD19/CD21 complex may modulate complement-dependent immune responses, and enhance the primary B cell response to antigens (10). In this model, CD21 may serve primarily as a ligand binding subunit, while CD19 could function as a signal-transducing subunit. However, it is possible that CD19 also has a specific ligand that has not yet been identified. The coupling of TAPA-1/CD81 to this multimeric protein complex was shown to be critical for CD19-mediated homotypic aggregation (11).
The relationship between CD19 and surface Ig signaling on mature B cells is controversial (12). Whereas one report indicated that anti-CD19 inhibited anti-IgM induced intracellular [Ca
2+] elevation (13), another report did not observe this effect (14). Anti-CD19 can inhibit mature B-cell proliferation induced by anti-IgM (13,15), but can also reduce the anti-Ig concentration needed to induce the proliferation of mature B cells (16). Although the aforementioned studies have suggested a role for CD19 in regulating Ig signaling in B cells, how CD19 functions in surface Ig negative B cell precursors is largely unknown. The absence of CD21 on B cell precursors (17) suggests that CD19 may interact with other molecules and/or subserve a potentially different signaling function on these cells.We have recently shown that cross-linking cell surface CD19 induces the homotypic aggregation of normal B cell precursors (18), indirectly suggesting that a signaling event can be transduced through CD19 on B cell precursors. We also found that the PTK inhibitor herbimycin A inhibits CD19-mediated homotypic aggregation (W.-K. Weng and T. W. LeBien, unpublished observations). This suggests that CD19-mediated homotypic aggregation is PTK- dependent, an observation previously reported by Kansas and Tedder (19).
In the current study, we have analyzed PTK activation following CD19 cross-linking in early human B-lineage cells, and have begun to characterize the substrates which undergo tyrosine phosphorylation. One of the signal transduction molecules selectively expressed in hematopoietic cells is the protein encoded by the vav proto-oncogene (20). Recent studies have shown that Vav is rapidly and transiently phosphorylated on tyrosine, and increases its Ras GTP/GDP exchange activity following T cell and B cell antigen receptor-initiated signal transduction (21,22). We now report that cross-linking CD19 on surface Ig negative B cell precursors and surface Ig positive immature B cells results in the rapid tyrosine phosphorylation of Vav, and a concomitant physical association of Vav with CD19 and PI 3-kinase. We further demonstrate that mitogen-activated protein (MAP) kinase-kinase (also known as MEK) and MAP kinase are activated following cross-linking of CD19. These results demonstrate that stimulation of CD19 can rapidly activate two major signaling pathways, suggesting a crucial signaling role for CD19 in antigen-independent and antigen-dependent stages of early B cell development.
MATERIALS AND METHODS
Cells
Human fetal bone marrow cells from normal 19-22 week gestational age fetuses were isolated as previously described (23). Briefly, the bone marrow cell suspension was centrifuged over a Ficoll-Hypaque gradient (Histopaque, Sigma Chemical Co., St. Louis, MO), and the interface cells were washed three times in RPMI-1640/2% fetal bovine serum (FBS) (Hyclone, Ogden, UT). CD10+/surface Ig light chain- B cell precursors were then purified from the interface cells by magnetic bead depletion of erythoid, myeloid, monocytoid, T-lineage and surface IgM+ B cells. The use of human tissue was in accordance with the guidelines established by the University of Minnesota Committee on the Use of Human Subjects in Research. BLIN-1 is a pre-B leukemic cell line (cytoplasmic m+, surface m+/surrogate light chain+, surface k/l-) originally established in this laboratory (24). 1E8 and lBLIN-1 are surface m/k+ and surface m/l+ subclones that spontaneously emerged from the parental BLIN-1 line. The leukemic B cells were maintained in RPMI 1640/5% FBS.
Antibodies
Hybridoma cells secreting anti-CD19 mAb (25C1) were generously donated by Dr. Stephen Peiper, University of Louisville School of Medicine, Louisville, KY. Hybridoma cells secreting anti-m heavy chain mAb (HB57), anti-k light chain mAb (HB61), anti-l light chain mAb (HP6054) and anti-class I MHC mAb (W6/32) were obtained from the American Type Culture Collection (Rockville, MD). Hybridoma cells secreting anti-b-tubulin mAb (E7) were obtained from the Developmental Studies Hybridoma Bank maintained by the Department of Pharmacology and Molecular Sciences, Johns Hopkins University School of Medicine, Baltimore, MD, and the Department of Biology, University of Iowa, Iowa City, IA, under contract NO1-HD-2-3144 from the National Institute of Child Health and Human Development. Monoclonal antibodies were initially purified from ascites by standard protein A-sepharose chromatography. Biotinylated antibodies were prepared by the method of Goding (25). Anti-surrogate light chain mAb (SLC-2) was kindly provided by Drs Kaiss Lassoued and Max Cooper, University of Alabama at Birmingham (26). Goat anti-mouse Ig and rabbit anti-mouse Ig were purchased from Southern Biotechnology Associates, Inc. (Birmingham, AL). Anti-phosphotyrosine (anti-p-Tyr) mAb (4G10), anti-MAP kinase mAb, anti-Vav Ab (IgG fraction of polyclonal rabbit antiserum for immunoprecipitation, monoclonal mouse ascites for immunoblotting) and anti-PI 3-kinase (p85) Ab (polyclonal rabbit antiserum) were purchased from Upstate Biotechnology Incorporated (Lake Placid, NY).
Flow Cytometry
The reagents and methods used to analyze cell surface expression of CD19 and Ig proteins by flow cytometry have been previously described (23). Briefly, 2 x 105 cells were stained with mAb against CD19 or biotinylated antibodies against Ig m heavy chain, k, l light chains and surrogate light chain. The binding of mAb was detected by secondary staining with goat anti-mouse Ig-fluorescein isothiocyanate (FITC) (Tago, Burlingame, CA) or streptavidin-phycoerythrin (PE) (Caltag, South San Francisco, CA). Cells were then fixed in 1% paraformaldehyde and analyzed on a FACScan (Becton Dickinson, San Jose, CA). Negative controls consisted of cells stained with equal concentrations of isotype-matched myeloma proteins or biotinylated myeloma proteins, followed by goat anti-mouse Ig-FITC or streptavidin-PE staining.
Cross-Linking Surface CD19 and Immunoblotting
Cells (1-1.5 x 106 from the indicated cell line or 2-3 x 106 normal B cell precursors) were incubated with anti-CD19 mAb (25C1) or other mAbs (all at 10 mg/ml) at 4 0C for 15 min, followed by cross-linking with goat anti-mouse Ig or rabbit anti-mouse Ig (both at 10 mg/ml) at 37 0C for the indicated times. (This procedure, which consists of incubating the primary antibody-stained cells with secondary antibody at 37 0C, will be referred as cross-linking throughout the manuscript.) The cells were then isolated by centrifugation and disrupted in lysis buffer containing 50 mM Tris pH 7.6, 300 mM NaCl, 5 mM EDTA and 0.5% Triton X-100 at 4 0C for 30 min. The lysis buffer also contained protease inhibitors (1 mg/ml aprotinin, 1 mg/ml leupeptin, 2 mM phenylmethylsulfonyl fluoride, 10 mM iodoacetamide, 25 mg/ml p-nitrophenyl-granidinobenzoate) and phosphatase inhibitors (2 mM sodium orthovanadate, 50 mM sodium fluoride). The soluble fraction was collected after centrifuging the whole cell lysate at 13,500xg at 4 0C for 30 min. The proteins were separated on a 11% SDS-PAGE gel and transferred onto a nitrocellulose membrane (Bio-Rad Laboratories, Hercules, CA). The membrane was blocked in Tris-buffered saline-Tween (TBST) (50 mM Tris pH 7.6, 150 mM NaCl, 0.1% Tween 20) containing 2% bovine serum albumin at 4 0C overnight prior to immunostaining. Detection of specific proteins was performed by incubating the membrane with the indicated primary antibodies (at concentrations suggested by the manufacturers: anti-p-Tyr mAb 0.75 mg/ml, monoclonal mouse anti-Vav ascites 1:1000, polyclonal rabbit anti-PI 3-kinase antiserum 1:1000, anti-MAP kinase mAb 2 mg/ml) at room temperature for 30 min, followed by secondary goat anti-mouse Ig-horseradish peroxidase or goat anti-rabbit Ig-horseradish peroxidase (Promega, Madison, WI) at room temperature for 30 min. The membrane was then developed using the enhanced chemiluminescence (ECL) detection system (Amersham Co., Arlington Heights, IL) to visualize the Ab-bound proteins. In some cases, the membrane was stripped in 62.5 mM Tris pH 6.7 containing 2% SDS, 100 mM b-mercaptoethanol at 50 0C for 30 min. The membrane was then blocked in TBST containing 2% bovine serum albumin, re-stained with the indicated primary antibodies and secondary horseradish peroxidase-conjugated antibodies, and re-developed using the ECL detection system. In the ECL system, the primary IgG heavy chains of the mAb used for cell cross-linking or immunoprecipitation are detected by secondary goat anti-mouse Ig-horseradish peroxidase, and shown as molecules of ~52 kDa in the immunoblots.
Immunoprecipitation
Cells (1 x 107) were cross-linked and lysed in 0.5% Triton X-100 lysis buffer as described above. The soluble fraction was collected after centrifuging twice at 13,500xg at 4 0C for 30 min. The cell lysates were then incubated with 5 mg of anti-p-Tyr mAb (4G10), 6 mg of rabbit anti-Vav, 4 ml of rabbit anti-PI 3-kinase (p85), or 10 mg of anti-CD19 mAb (25C1) at 4 0C overnight. Immune complexes were precipitated with protein A-Sepharose beads (Sigma, St. Louis, MO) or rabbit anti-mouse Ig-coated protein A-Sepharose beads at 4 0C for 2 h. The beads were washed four times in lysis buffer. The precipitated proteins were eluted by boiling the beads in sample buffer (80 mM Tris pH 6.8, 3% SDS, 15% glycerol, 0.01% bromophenol blue, 5% b-mercaptoethanol). In some experiments, tyrosine-phosphorylated proteins were eluted from the washed beads using 10 mM p-nitro-phenyl phosphate (pNPP) at 4 0C for 30 min (27).
MEK (MAP Kinase Kinase) Activity Assay
A glutathione s-transferase-[K71A] erk1 fusion protein (Upstate Biotechnology Incorporated, Lake Placid, NY), which will be referred to as GST-MAP kinase throughout the manuscript, was used as the substrate for MEK in an in-vitro kinase assay. This GST-MAP kinase was constructed by fusing full-length human MAP kinase (with site-directed mutagenesis to convert Lys-71 to Ala) to glutathione s-transferase protein. Mutation of Lys-71 to Ala in GST-MAP kinase results in a catalytically inactive version of MAP kinase that cannot autophosphorylate nor phosphorylate other proteins (28). In this assay, cells (1 x 107) were cross-linked, lysed in 0.5% Triton X-100 lysis buffer, and soluble fractions isolated as described above. Ten mM MgCl2, 20 mM 3-N-morpholino propanesulfonic acid (MOPS) pH 7.2 and 5 mg of GST-MAP kinase agarose conjugate were added to the cell lysates, and an in-vitro kinase reaction was initiated by the addition of 5 mM ATP and 10 mCi [g-32P]ATP (4500 Ci/m mol) (ICN Biomedicals, Inc., Costa Mesa, CA). The reaction was performed at room temperature for 10 min and stopped by adding ice-cold 1 mM cold ATP. The agarose beads were then washed five times in 0.5% Triton X-100 lysis buffer. The GST-MAP kinase was eluted by boiling in sample buffer for 4 min, and then separated on a 11% SDS-PAGE gel. The gel was dried and developed by autoradiography. In some experiments, the 32P incorporation was quantitated by densitometric scanning of the autoradiographs. This was accomplished using a UC630 MaxColor scanner (UMAX Data System Inc., Hsinchu, Taiwan) connected to a Macintosh computer with the NIH Image (version 1.55) program. The density values were determined for each lane and normalized to values obtained from the Coomassie blue-stained gel in the same experiment.
RESULTS
Cross-Linking CD19 Activates PTK in B Cells at Distinct Developmental Stages
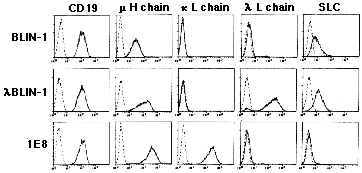
The BLIN-1 cell line and its differentiated subclones are potentially interesting cellular tools for examining the influence of CD19 cross-linking on PTK activation. Figure 1 shows the expression of CD19 and Ig proteins on the pre-B acute lymphoblastic leukemia cell line BLIN-1 and its differentiated immature B cell subclones lBLIN-1 and 1E8. BLIN-1 expresses low levels of m heavy chain and surrogate light chain, but does not express k or l light chains. 1E8 and lBLIN-1 express higher levels of m heavy chains and k and l light chains, respectively. Importantly, the three cell lines expressed identical levels of cell surface CD19.

Figure 1.
Expression of CD19 and Ig proteins on the surface of the pre-B cell line BLIN-1 and immature B cell lines lBLIN-1 and 1E8 Histograms define fluorescence intensity on the X axis and relative cell number on the Y axis. Dashed lines represent background staining. Solid lines represent specific staining by respective mAb.
We first examined whether CD19 cross-linking would increase PTK activity and subsequent protein tyrosine phosphorylation in 1E8 cells. As shown in Fig. 2A, cross-linking CD19 or surface IgM
for 1 or 5 min increased PTK activity and tyrosine phosphorylation of multiple protein substrates, compared to control unstimulated cells (Cont.) or cells incubated with goat anti-mouse Ig alone (20 Ab). Very slight PTK activation occurred in cells incubated with IgG1 myeloma control or anti-class I MHC mAb. Cross-linking surface IgM resulted in rapid and strong tyrosine phosphorylation of substrates with an approximate molecular mass of 55, 62, 65, 72, 80, 95, 110, 125, 140, and 180 kDa. Cross-linking CD19 resulted in tyrosine phosphorylation of a smaller number of substrates of 62, 95, 110, 125, and 140 kDa. The strong tyrosine phosphorylated 72 kDa molecule in IgM cross-linking cells is probably Syk (29). Anti-b-tubulin immunoblotting demonstrated that similar amounts of protein were electrophoresed in each lane. Time-course analysis of PTK activation in 1E8 revealed a peak response between 0.5 and 1 min that gradually dissipated over 30 min in both CD19 and IgM cross-linked cells (Fig. 2B). A tyrosine-phosphorylated 43 kDa molecule was detected in both IgM and CD19 cross-linked 1E8 when the blot was exposed for a longer time (data not shown). Similarly, cross-linking CD19 on BLIN-1 and lBLIN-1 activated PTK with similar kinetics to those shown for 1E8 cells in Fig. 2 (data not shown and see below).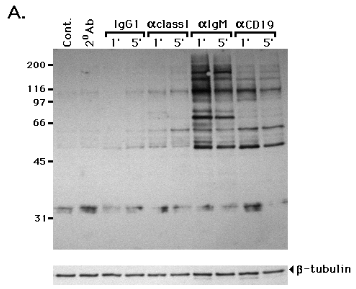
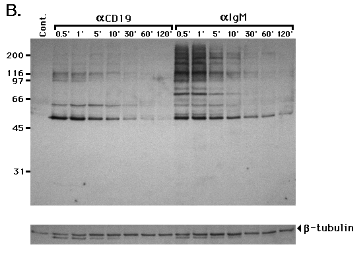
Figure 2.
Cross-linking CD19 and Ig m heavy chain increases PTK activity 1E8 cells were incubated with control mouse IgG1 (IgG), anti-class I MHC (aclass I), anti-m heavy chain (aIgM) or anti-CD19 (aCD19) at 4 0C for 15 min, and further cross-linked with goat anti-mouse Ig at 37 0C for 1 or 5 min. Controls included unstimulated cells (Cont.) or cells only incubated with goat anti-mouse Ig at 37 0C for 5 min (20 Ab). The cells were then lysed and processed for anti-p-Tyr immunoblotting (A.). The same blot was stripped and re-stained with anti-b-tubulin mAb to confirm that equal amounts of protein were electrophoresed in each lane (lower strip in A.). Time course analysis of PTK activation in 1E8 incubated with anti-CD19 (aCD19) or anti-m heavy chain (aIgM) at 4 0C for 15 min, and cross-linked at 37 0C for 0.5 to 120 min. Cont.= unstimulated cells. The cells were then lysed and processed for anti-p-Tyr immunoblotting (B.). The blot was stripped and re-stained with anti-b-tubulin (lower strip in B.). In blots A. and B., the ~52 kDa molecule detected by anti-p-Tyr immunoblotting is probably the primary IgG heavy chain of the mAb used for cell cross-linking. This possibility was confirmed by staining the membrane with secondary goat anti-mouse Ig-horseradish peroxidase only, and developing in the ECL system (data not shown). This cross reaction was also evident in the anti-b-tubulin blots as a protein migrating slightly faster than the 55 kDa b-tubulin. Molecular mass standards are expressed as kDa on the Y axis.
The Vav Proto-oncoprotein Undergoes Tyrosine Phosphorylation Following CD19 Cross-Linking
The 95 kDa proto-oncoprotein Vav (20), a putative guanine nucleotide exchange factor, becomes tyrosine phosphorylated following cross-linking of surface Ig (22) or the T cell receptor (21). Data in Fig. 2 revealed considerable overlap in the size of the tyrosine phosphorylated substrates following cross-linking of surface IgM and CD19, including a 95 kDa protein. We therefore examined whether Vav became tyrosine phosphorylated following CD19 cross-linking. 1E8 and BLIN-1 were cross-linked with anti-CD19 or anti-class I MHC, lysed, immunoprecipitated with anti-p-Tyr, and immunoblotted with anti-Vav.

Figure 3.
Vav is present in anti-p-Tyr immunoprecipitates following CD19 cross-linking 1E8 and BLIN-1 were incubated with control IgG1 (Cont.), anti-CD19 (aCD19), or anti-class I MHC (aclass I) at 4 0C for 15 min, and cross-linked at 37 0C for 1 or 5 min. The cells were then lysed and immunoprecipitated with control IgG or anti-p-Tyr. The precipitated proteins were separated by SDS-PAGE, transferred to a nitrocellulose membrane, and immunoblotted with anti-Vav. The non-specific bands between 45 and 65 kDa represent Ig heavy chains used for cell cross-linking or immunoprecipitation, or other proteins stained by secondary horseradish peroxidase-conjugated Ab in the ECL detection system.As shown in Fig. 3, Vav was detected in anti-p-Tyr immunoprecipitates following 1 and 5 min of CD19 cross-linking in 1E8 and BLIN-1, whereas anti-p-Tyr immunoprecipitates of class I MHC cross-linked cells gave very weak Vav staining after 5 min in BLIN-1 only. Neither control IgG1 cross-linking nor control IgG1 immunoprecipitation revealed any detectable Vav. This result suggests that cross-linking CD19 leads to PTK activation and subsequent tyrosine phosphorylation of Vav. Alternatively, Vav may associate with some tyrosine phosphorylated protein following CD19 cross-linking, and be co-precipitated with anti-p-Tyr mAb. To distinguish between these two possibilities, anti-Vav immunoprecipitates of cell lysates from CD19 cross-linked 1E8 and BLIN-1 were analyzed by anti-p-Tyr immunoblotting. As shown in Fig. 4A, an increase in tyrosine phosphorylation of the 95 kDa Vav was detected at 0.5 min and largely disappeared 5 min after CD19 cross-linking. When this blot was stripped and re-stained with anti-Vav, identical levels of Vav protein were present in all lanes (Fig. 4B). Therefore, the results in Fig. 4A cannot be ascribed to potential differences in the amount of immunoprecipitated Vav electrophoresed in each lane. In a separate experiment, 1E8 and BLIN-1 were cross-linked with anti-CD19, lysed, and subjected to rigorous pre-clearing by three rounds of immunoprecipitation with anti-CD19 or control IgG1. The lysates were then immunoprecipitated with anti-Vav and subjected to anti-p-Tyr immunoblotting. There was no detectable decrease in the intensity of the 95 kDa tyrosine phosphorylated Vav followed pre-clearing CD19, compared to pre-clearing with control IgG1 (data not shown). Thus, it is unlikely that the 95 kDa tyrosine phosphorylated protein in Fig. 4A is CD19.
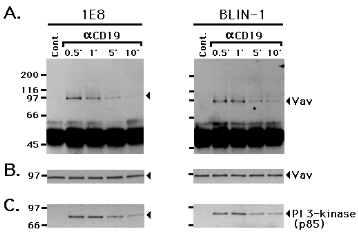
Figure 4.
Vav undergoes tyrosine phosphorylation following CD19 cross-linking 1E8 and BLIN-1 were incubated with control IgG1 (Cont.), or anti-CD19 (aCD19) at 4 0C for 15 min, and cross-linked at 37 0C for 0.5 to 10 min. The cells were lysed and immunoprecipitated with anti-Vav. The precipitated proteins were then separated by SDS-PAGE, transferred to a nitrocellulose membrane, and immunoblotted with anti-p-Tyr (A.). The blot was then stripped, and re-stained with anti-Vav (B.) or anti-PI 3-kinase (C.).
Cross-Linking CD19 Induces an Interaction Between Vav and PI 3-kinase
Since Tuveson et al. reported that PI 3-kinase can bind to phosphorylated CD19 (7), we examined whether Vav interacts with the PI 3-kinase following CD19 cross-linking. Fig. 4C shows that the p85 subunit of PI 3-kinase was detected in the anti-Vav immunoprecipitates following 0.5 min of CD19 cross-linking in 1E8 and BLIN-1, whereas no PI 3-kinase was detected in anti-Vav immunoprecipitates from control IgG1 treated cells.
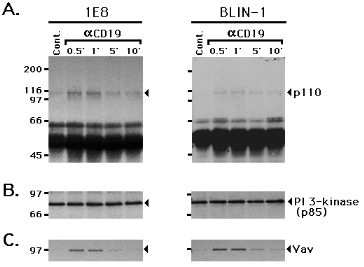
Figure 5.
Cross-linking CD19 induces an interaction between Vav and PI 3-kinase 1E8 and BLIN-1 were incubated with control IgG1 (Cont.) or anti-CD19 (aCD19) at 4 0C for 15 min, and cross-linked at 37 0C for 0.5 to 10 min. The cells were lysed and immunoprecipitated with anti-PI 3-kinase. The precipitated proteins were then separated by SDS-PAGE, transferred to a nitrocellulose membrane, and immunoblotted with anti-p-Tyr (A.). The blot was then stripped, and re-stained with anti-PI 3-kinase (B.) or anti-Vav (C.).To more directly examine the potential interaction between Vav and PI 3-kinase, CD19 was cross-linked on 1E8 and BLIN-1 and cell lysates were immunoprecipitated with anti-PI 3-kinase (p85). As shown in Fig. 5A, anti-PI 3-kinase immunoprecipitates blotted with anti-p-Tyr revealed a 110 kDa molecule of unknown identity, which may be the catalytic subunit of PI 3-kinase. Control staining with anti-PI 3-kinase revealed that comparable amounts of immunoprecipitated protein were electrophoresed in each lane (Fig. 5B). Fig. 5C shows the same blot re-stained with anti-Vav, demonstrating that Vav was co-precipitated with PI 3-kinase in CD19 cross-linked cells. Although Vav was tyrosine phosphorylated following CD19 cross-linking (Fig. 4A), no tyrosine phosphorylation of co-precipitated Vav was detected (Fig. 5A). Tyrosine phosphorylation of the p85 subunit of PI 3-kinase has been observed following receptor-initiated signal transduction in lymphocytes (30,31). However, no tyrosine phosphorylation of p85 was detected following CD19 cross-linking (Fig. 5A).
Cross-Linking CD19 Induces an Interaction Between Vav and CD19
Bustelo et al. reported that activation of the epidermal growth factor receptor or platelet-derived growth factor receptor leads to tyrosine phosphorylation of Vav, and Vav becomes physically coupled to the receptor through its SH2 domain (32). We therefore examined whether CD19 cross-linking induced an interaction between Vav and CD19. As shown in Fig. 6A, Vav was detected in the anti-CD19 immunoprecipitates following 1 min of CD19 cross-linking in 1E8 and BLIN-1, whereas anti-CD19 immunoprecipitates of control IgG1 treated cells only revealed weak staining of Vav. In a similar experiment, only weak staining of Vav (comparable to that in control IgG1 treated cells) was detected in the anti-CD19 immunoprecipitates of anti-class I MHC cross-linked 1E8 and BLIN-1 cells (data not shown). Interestingly, this increase in the Vav/CD19 interaction peaked at 1 min and decreased by 5 min, which correlated with the degree of tyrosine phosphorylation of Vav (Fig. 4A). There was no detectable Vav in control IgG1 immunoprecipitates of IgG1 treated cells. However, some Vav was detected in control IgG1 immunoprecipitates of anti-CD19 cross-linked cells. Since some CD19/Vav may complex to anti-CD19 used for cross-linking, it is likely that these immune complexes were precipitated by rabbit anti-mouse Ig-coated protein A-Sepharose beads during the immunoprecipitation procedure. The 185 and 105 kDa proteins present in the control IgG1 immunoprecipitates are non-specific proteins that were stained by secondary horseradish peroxidase-conjugated Ab in the ECL detection system. In Fig. 6B, the p85 subunit of PI 3-kinase was detected in anti-CD19 immunoprecipitates of CD19 cross-linked cells, whereas little PI 3-kinase was detected in control IgG1 treated cells, consistent with the report by Tuveson and colleagues (7). The appearance of some PI 3-kinase in the control IgG1 immunoprecipitates of CD19 cross-linked cells can probably be explained by rabbit anti-mouse precipitation of anti-CD19 immune complexes, as discussed above.
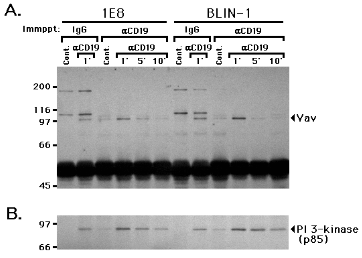
Figure 6.
Cross-linking CD19 induces an interaction between Vav and CD19 1E8 and BLIN-1 were incubated with control IgG1 (Cont.) or anti-CD19 (aCD19) at 4 0C for 15 min, and cross-linked at 37 0C for 1, 5 or 10 min. The cells were lysed and immunoprecipitated with control IgG1 or anti-CD19. The precipitated proteins were then separated by SDS-PAGE, transferred to a nitrocellulose membrane, and immunoblotted with anti-Vav (A.). The blot was then stripped and re-stained with anti-PI 3-kinase (B.).
MAP Kinase Activation Following CD19 Cross-Linking
When the BLIN-1 pre-B cell line and the
lBLIN-1 and 1E8 immature B cell lines were cross-linked with anti-CD19, virtually identical patterns of tyrosine phosphorylation were detected in lBLIN-1 and 1E8. BLIN-1, on the other hand, exhibited some qualitative differences compared to lBLIN-1 and 1E8 (Fig. 7A). One notable difference was the presence of an approximately 43 kDa phosphotyrosine protein in lBLIN-1 and 1E8, that was absent in BLIN-1. Since p42 MAP kinase is phosphorylated on tyrosine and threonine, and exhibits mobility retardation in SDS-PAGE following growth factor receptor or surface IgM cross-linking (33,34), we reasoned that the 43 kDa protein may be activated p42 MAP kinase.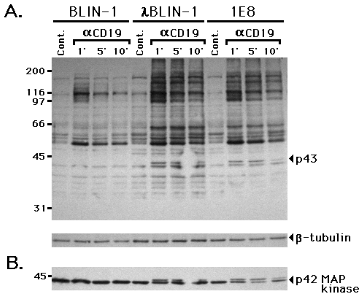
Figure 7.
MAP kinase activation following CD19 cross-linking in immature B cell lines BLIN-1, lBLIN-1 and 1E8 were incubated with control IgG1 (Cont.) or anti-CD19 (aCD19) at 4 0C for 15 min, and cross-linked at 37 0C for 1, 5 or 10 min. The cells were lysed, electrophoresed, and immunoblotted with anti-p-Tyr (A.). The blot was then stripped, and re-stained with anti-b-tubulin (lower strip in A.) or anti-MAP kinase (B.).To examine this possibility, we stripped the membrane in Fig. 7A and re-stained with anti-MAP kinase mAb. As shown in Fig. 7B, anti-MAP kinase staining revealed a 42 kDa protein in all lanes, irrespective of CD19 cross-linking. However, a slightly less mobile molecule of approximately 43 kDa was detected following CD19 cross-linking in
lBLIN-1 and 1E8, but not BLIN-1. The 43 kDa protein in Fig. 7B precisely overlaid the 43 kDa protein detected by anti-p-Tyr immunoblotting in Fig. 7A. Fig. 8 shows that the mobility retardation of p42 MAP kinase was detected as early as 0.5 min following CD19 cross-linking, peaked at 10 min, and dramatically subsided by 30 min.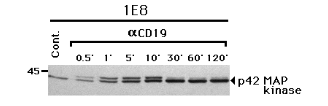
Figure 8.
Time course analysis of the mobility retardation of p42 MAP kinase following CD19 cross-linking Unstimulated 1E8 cells (Cont.) or 1E8 were incubated with anti-CD19 (aCD19) at 4 0C for 15 min and cross-linked at 37 0C for 0.5 to 120 min. The cells were then lysed, electrophoresed, and immunoblotted with anti-MAP kinase (p42).We then examined the effect of CD19 cross-linking on normal B cell precursors isolated from human fetal bone marrow. As shown in Fig. 9A, anti-p-Tyr immunoblotting revealed a modest increase in tyrosine phosphorylation of substrates of 55, 62, 95, 100, 110 and 125 kDa following CD19 cross-linking. When the blot was stripped and re-stained with anti-MAP kinase, no mobility retardation of p42 MAP kinase was detected following CD19 cross-linking (Fig. 9B). In contrast, phorbol 12-myristate 13-acetate (PMA) treatment resulted in faint tyrosine phosphorylation of a 43 kDa molecule in the anti-p-Tyr immunoblot (Fig. 9A), and induced pronounced mobility retardation of p42 MAP kinase (Fig. 9B). PMA treatment also induced mobility retardation of p42 MAP kinase in the pre-B cell line BLIN-1 (data not shown).
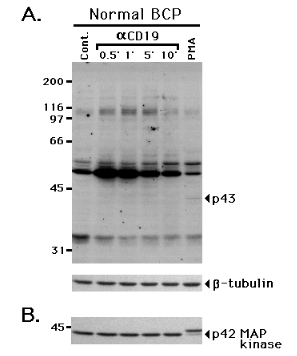
Figure 9.
PMA, but not CD19 cross-linking, activates MAP kinase in normal B cell precursors Surface CD10+/m- normal B cell precursors were incubated with control IgG1 (Cont.) or anti-CD19 (aCD19) at 4 0C for 15 min, and cross-linked at 37 0C for 1, 5 or 10 min. For PMA treatment, the cells were incubated with 10 ng/ml PMA at 37 0C for 10 min. The cells were lysed, electrophoresed, and immunoblotted with anti-p-Tyr (A.). The blot was then stripped, and re-stained with anti-b-tubulin (lower strip in A.) or anti-MAP kinase (B.).
MEK Activation Following CD19 Cross-Linking
In order to more directly analyze activation of the MAP kinase pathway, we assayed for MEK activation following CD19 cross-linking using an in vitro kinase assay to assess the capacity of MEK to phosphorylate MAP kinase. Since MAP kinase is the only known substrate of MEK (35), a GST-MAP kinase fusion protein was used as a substrate in the in vitro kinase assay. As shown in Fig. 10, 1E8 and BLIN-1 cells treated with control IgG1 exhibited low MEK activity, based on the weakly phosphorylated GST-MAP kinase fusion protein. Phosphorylation of GST-MAP kinase was slightly enhanced by cross-linking class I MHC for 1 min, and more intensely enhanced by cross-linking CD19 for 1 or 5 min in both 1E8 and BLIN-1. Scanning densitometry was used to quantify the autoradiographic signals in Fig. 10. Anti-class I MHC cross-linking of 1E8 cells led to a 2.0-fold increase in GST-MAP kinase phosphorylation, and anti-CD19 cross-linking led to a 3.4-fold and 3.7-fold increase in GST-MAP kinase phosphorylation at 1 min and 5 min, respectively. Similarly, anti-class I MHC cross-linking of BLIN-1 cells led to a 1.8-fold increase in GST-MAP kinase phosphorylation, and anti-CD19 cross-linking led to a 2.4-fold and 3.4-fold increase in GST-MAP kinase phosphorylation at 1 min and 5 min, respectively.
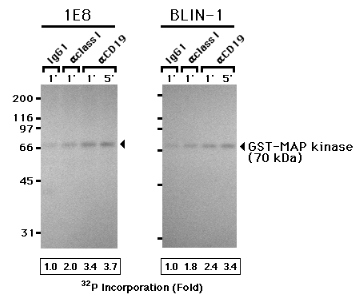
Figure 10.
Cross-linking CD19 activates MEK in pre-B and immature B cell lines 1E8 and BLIN-1 were incubated with control IgG1 (IgG1), anti-class I MHC (aclass I), or anti-CD19 (aCD19) at 4 0C for 15 min, and cross-linked at 37 0C for 1 or 5 min. The cells were then lysed, and subjected to an in vitro kinase reaction using GST-MAP kinase agarose conjugates and [g-32P] ATP. The GST-MAP kinase was then separated by SDS-PAGE. The gel was dried and processed for autoradiography. A representative autoradiograph is shown and the arrow points to the 70 kDa GST-MAP kinase. Quantitation of 32P incorporation into the GST-MAP kinase was accomplished by densitometric scanning of the autoradiograph. The density values were determined for each lane and normalized to values obtained from the Coomassie blue-stained gel in the same experiment. The normalized values from control IgG1 cross-linked cells were assigned a value of 1.0. The values obtained from anti-class I and anti-CD19 cross-linking were then expressed as fold increase above the control IgG1.
DISCUSSION
The human B cell-specific cell surface molecule CD19 was identified and characterized over 10 years ago (1), and its complete amino acid sequence has been deduced from a cDNA (3). However, a putative CD19 ligand has not been described, and functional analyses have therefore relied on anti-CD19 mAb. Most of the functional studies on CD19 have focused on the relationship between CD19 and surface Ig signaling on B cells. Several reports have shown that anti-CD19 can inhibit the proliferation of B cells induced by anti-IgM (13-15). Other studies have demonstrated that anti-CD19 can enhance or inhibit, depending on the nature of the signaling event being modified or the identity of the responding cell (36,37). In contrast, using murine L cells transfected with the CD32 Fc receptor to present anti-CD19 or anti-IgM on the L cell surface, Carter et al. demonstrated that co-ligation of surface CD19 and Ig can reduce the Ig concentration required to induce the proliferation of mature B cells (16). Anti-CD19 presented on the L cells may mimic a putative cell bound CD19 ligand by delivering a potent cross-linking stimulus through CD19.
CD19 is expressed on B cell precursors at levels comparable to mature B cells (2,3). However, the function of CD19 on surface Ig negative B cell precursors has not been as thoroughly investigated. We have recently shown that IL-7 increases the level of cell surface CD19, and cross-linking CD19 triggers the homotypic aggregation of normal B cell precursors (18). We proposed (18) that anti-CD19 mAb 25C1 may activate a functional domain in a manner analogous to the putative CD19 ligand. We also found that the PTK inhibitor herbimycin A disrupted CD19-mediated homotypic aggregation in B cell precursors (W.-K. Weng and T.W. LeBien, unpublished observations), implicating PTK activation in CD19 signaling.
As an extension of our prior studies (18), we herein examined the activation of PTK following cross-linking of CD19. We observed PTK activation on both pre-B and immature B cell lines, as well as normal B cell precursors, which led to tyrosine phosphorylation of multiple electrophoretically distinct substrates in all cases (Figs. 2,7, and 9). Separate cross-linking of CD19 and IgM on the 1E8 immature B cell line activated PTK with similar kinetics, and revealed tyrosine phosphorylated proteins of similar sizes (Fig. 2). Comparable results have been reported using other B-lineage cells (38,39). This data suggests that PTK activated following cross-linking of CD19 and surface IgM may have common substrates. In this regard, CD19 undergoes tyrosine phosphorylation following cross-linking of both surface IgM and CD19 (7,38,39). We have now identified one of the tyrosine phosphorylated substrates in CD19 cross-linked cells as Vav, which has recently been demonstrated to undergo rapid and transient tyrosine phosphorylation following B cell antigen receptor-activated signal transduction (22). In both pre-B and immature B cell lines Vav was tyrosine phosphorylated within 1 min after CD19 cross-linking (Fig. 4A). However, the amount of Vav detected in anti-p-Tyr immunoprecipitates remained steady or slightly increased between 1 and 5 min after CD19 cross-linking (Fig.3), whereas tyrosine phosphorylation of Vav substantially decreased between 1 and 5 min after CD19 cross-linking (Fig. 4A). This suggests that in addition to the tyrosine phosphorylation of Vav, Vav may also associate with tyrosine phosphorylated proteins (which could include other proteins in addition to CD19) following CD19 cross-linking. Our results establish a potentially important link between CD19 cross-linking and tyrosine phosphorylation of Vav, which has implications for activation of the Ras pathway (see below). It is also possible that a Ras-independent pathway may be activated through Vav. This possibility receives support from the knowledge that Vav has multiple (potential) functional domains, including an amino-terminal helix-loop-helix/leucine zipper motif (40,41) and two nuclear localization signals (42).
One of the most important observations in the current study was the discovery of a potentially complex physical interaction between CD19, Vav, and the p85 subunit of PI 3-kinase. Cross-linking CD19 leads to the association of tyrosine phosphorylated CD19 with p85 via two Tyr-X-X-Met motifs in the CD19 cytoplasmic tail (7). This interaction serves to activate the p110 catalytic subunit of PI 3-kinase (7), although the mechanism of activation is unknown. The CD19/p85 association was also predicted based on the use of phosphopeptide libraries to determine the peptide-binding site specificity of SH2 domains (5). Our data in Fig. 6B are consistent with a CD19/p85 physical association enhanced by CD19 cross-linking. This CD19/p85 interaction depends on the tyrosine phosphorylation of CD19 (7), therefore, it can also be used as an indirect indication of tyrosine phosphorylation of CD19. Since the CD19/p85 interaction was detected after 1 min CD19 cross-linking and started to decrease by 10 min, this suggests that CD19 was tyrosine-phosphorylated between 1 and 10 min following cross-linking. Furthermore, the collective data in Figs. 4 and 5 provide strong evidence that CD19 cross-linking induces an increase in the physical interaction between p85 and Vav. This activation induced interaction between p85 and Vav has, to our knowledge, not been described. Our studies did not delineate the mechanism by which Vav and p85 interact. However, the proline rich region of the p85 subunit can interact with SH3 domains of Fyn and Lyn, and this interaction increases the specific activity of PI 3-kinase (43). It remains to be determined whether either of the two SH3 domains in Vav can interact with the proline rich region in p85. Interaction via the Vav SH2 domain seems unlikely, since no detectable tyrosine phosphorylation of p85 occurred following CD19 cross-linking (Fig. 5A). Whereas CD19 cross-linking induced the tyrosine phosphorylation of Vav (Fig. 4A), no tyrosine phosphorylation of p85-associated Vav was detected (Fig. 5A). There are two possible explanations for the latter result. First, only non-phosphorylated Vav may associate with p85 following CD19 cross-linking. Second, only a fraction of p85-associated Vav may become tyrosine phosphorylated, which may be difficult to detect by anti-p-Tyr immunoblotting.
In addition to the p85/CD19 and p85/Vav interactions, we also observed that CD19 cross-linking enhanced the interaction between Vav and CD19 (Fig. 6). This interaction could be mediated through the SH2 domain of Vav, since a Tyr-Glu-Glu-Pro motif in CD19 has been proposed to specifically bind to the Vav SH2 domain (6). It is also possible that the intereaction between Vav and CD19 is an indirect one via a third protein, e.g. the p85 subunit of PI 3-kinase. Lyn and Lck Src-family PTK have been reported to associate with CD19, and their enzymatic activities increase following CD19 cross-linking (38,44). This enhanced interaction between CD19 and Vav may facilitate the physical translocation of Vav into close proximity to activated PTK, thereby stimulating the tyrosine phosphorylation and subsequent activation of Vav GTP/GDP exchange activity. However, it is not known whether Lyn, Lck, or other Src-family PTK phosphorylate Vav.
Conversion of inactive GDP-bound Ras to active GTP-bound Ras is a critical switch in signal transduction that culminates in activation of the MAP kinase pathway. Recent evidence supports a model in which a primary role of GTP-bound Ras in the proximal stages of the MAP kinase pathway is to localize c-Raf to the plasma membrane, thereby activating c-Raf (45,46). Activated c-Raf then phosphorylates MEK, which in turn activates MAP kinase by dual phosphorylation on Tyr and Thr residues (47). Since tyrosine phosphorylation of Vav enhances its Ras GTP/GDP exchange activity (21,22), we tested whether the MAP kinase pathway was activated secondary to CD19 cross-linking. This was investigated using two methods. The first took advantage of the fact that activation of MAP kinase correlates with a shift in the electrophoretic mobility of MAP kinase to a less mobile form (33,34). Cross-linking CD19 on the immature B cell lines 1E8 and
lBLIN-1 induced a clear shift in the mobility of p42 MAP kinase (Fig. 7) but, surprisingly, this did not occur on the BLIN-1 pre-B cell line (Fig. 7) or normal B cell precursors (Fig. 9). However, stimulation of normal B cell precursors (Fig. 9) and BLIN-1 (data not shown) with PMA did activate p42 MAP kinase by the criterion of a shift in electrophoretic mobility. We more directly analyzed activation of the MAP kinase pathway by assaying the capacity of MEK to phosphorylate MAP kinase. As shown in Fig. 10, CD19 cross-linking induced detectable MEK activation in both pre-B (BLIN-1) and immature B (1E8) cell lines. It is conceivable that an increase in MEK catalytic activity and subsequent MAP kinase activation may be independently regulated in pre-B cells. Our collective results are consistent with Ras pathway activation following CD19 cross-linking. Furthermore, antigen induced B cell stimulation can activate the Ras pathway (48). It would appear, therefore, that signaling through surface Ig and CD19 may both trigger Ras pathway activation through a step involving the tyrosine phosphorylation and activation of Vav.In conclusion, we have demonstrated that surface Ig negative and surface Ig positive human B-lineage cells respond to CD19 cross-linking by PTK activation and formation of a physical complex (of unknown stoichiometry) consisting of CD19, Vav, and PI 3-kinase. One interpretation of this data would hold that two distinct signaling pathways are activated through CD19, one involving Vav/Ras/MAP kinase, and the other involving PI 3-kinase. Future studies will need to determine the changes in gene expression that occur in B-lineage cells as a consequence of these pathways being activated, and the ultimate effect of these events on the survival, growth, and differentiation of early B-lineage cells.
REFERENCES
1. Nadler, L.M., Anderson, K.C., Marti, G., Bates, M., Park, E., Daley, J.F., and Schlossman, S.F. (1983) B4, a human B lymphocyte-associated antigen expression on normal, mitogen-activated, and malignant B lymphocytes. J. Immunol. 131:244-250
2. Uckun, F.M., Jaszcz, W., Ambrus, J.L., Fauci, A.S., Gajl-Peczalska, K., Song, C.W., Wick, M.R., Myers, D.E., Waddick, K., and Ledbetter, J.A. (1988) Detailed studies on expression and function of CD19 surface determinant by using B43 monoclonal antibody and the clinical potential of anti-CD19 immunotoxins. Blood 71:13-29
3. Tedder, T.F., and Isaacs, C.M. (1989) Isolation of cDNAs encoding the CD19 antigen of human and mouse B lymphocytes: a new member of the immunoglobulin superfamily. J. Immunol. 143:712-717
4. Zhou, L.J., Ord, D.C., Hughes, A.L., and Tedder, T.F. (1991) Structure and domain organization of the CD19 antigen of human, mouse and guinea pig B lymphocytes. Conservation of the extensive cytoplasmic domain. J. Immunol. 147:1424-1432
5. Songyang, Z., Shoelson, S.E., Chaudhuri, M., Gish, G., Pawson, T., Haser, W.G., King, F., Roberts, T., Ratnofsky, S., Lechleider, R.J., Neel, B.G., Birge, R.B., Fajardo, J.E., Chou, M.M., Hanafusa, H., Schaffhausen, B., and Cantley, L.C. (1993) SH2 domains recognize specific phosphopeptide sequences. Cell 72:767-778
6. Songyang, Z., Shoelson, S.E., McGlade, J., Olivier, P., Pawson, T., Bustelo, X.R., Barbacid, M., Sabe, H., Hanafusa, H., Yi, T., Ren, R., Baltimore, D., Ratnofsky, S., Feldman, R.A., and Cantley, L.C. (1994) Specific motifs recognized by the SH2 domains of Csk, 3BP2, fps/fes, GRB-2, HCP, SHC, Syk, and Vav. Mol. Cell. Biol. 14:2777-2785
7. Tuveson, D.A., Carter, R.H., Soltoff, S.P., and Fearon, D.T. (1993) CD19 of B cells as a surrogate kinase insert region to bind phosphatidylinositol 3-kinase. Science 260:986-988
8. Matsumoto, A.K., Kopicky-Burd, J., Carter, R.H., Tuveson, D.A., Tedder, T.F., and Fearon, D.T. (1991) Intersection of the complement and immune systems: a signal transduction complex of the B lymphocyte-containing complement receptor type 2 and CD19. J. Exp. Med. 173:55-64
9. Bradbury, L.E., Kansas, G.S., Levy, S., Evans, R.L., and Tedder, T.F. (1992) The CD19/CD21 signal transducing complex of human B lymphocytes includes the target of antiproliferative antibody-1 and Leu-13 molecules. J. Immunol. 149:2841-2850
10. Hebell, T., Ahearn, J.M., and Fearon, D.T. (1991) Suppression of the immune response by a soluble complement receptor of B lymphocytes. Science 254:102-105
11. Matsumoto, A.K., Martin, D.R., Carter, R.H., Klickstein, L.B., Ahearn, J.M., and Fearon, D.T. (1993) Functional dissection of the CD19/CD21/TAPA-1/Leu-13 complex of B lymphocytes. J. Exp. Med. 178:1407-1417
12. van Noesel, C.J.M., Lankester, A.C., and van Lier, R.A.W. (1993) Dual antigen recognition by B cells. Immunol. Today 14:8-11
13. Pezzutto, A., Dorken, B., Rabinovitch, P.S., Ledbetter, J.A., Moldenhauer, G., and Clark, E.A. (1987) CD19 monoclonal antibody HD37 inhibits anti-immunoglobulin-induced B-cell activation and proliferation. J. Immunol. 138:2793-2799
14. de Rie, M.A., Schumacher, T.N.M., van Schijndel, G.M., van Lier, R.A.W., and Miedema, F. (1989) Regulatory role of CD19 molecules in B cell activation and differentiation. Cell Immunol. 118:368-381
15. Barrett, T.B., Shu, G.L., Draves, K.E., Pezzutto, A., and Clark, E.A. (1990) Signaling through CD19, Fc receptor or transforming growth factor-
b: each inhibits the activation of resting human B cells differently. Eur. J. Immunol. 20:1053-105916. Carter, R.H., and Fearon, D.T. (1992) CD19: lowering the threshold for antigen receptor stimulation of B lymphocytes. Science 256:105-107
17. LeBien, T.W., Wormann, B., Villablanca, J.G., Law, C.L., Steinberg, L.M., Shah, V.O., and Loken, M.R. (1990) Multiparameter flow cytometric analysis of human fetal bone marrow B cells. Leukemia 4:354-358
18. Wolf, M.L., Weng, W.-K., Stieglbauer, K.T., Shah, N., and LeBien, T.W. (1993) Functional effect of IL-7 enhanced CD19 expression of human B cell precursors. J. Immunol. 151:138-148
19. Kansas, G.S., and Tedder, T.F. (1991) Transmembrane signals generated through MHC class II, CD19, CD20, CD39, and CD40 antigens induce LFA-1-dependent and independent adhesion in human B cells through a tyrosine kinase-dependent pathway. J. Immunol. 147:4094-4102
20. Katzav, S., Martin-Zanca, D., and Barbacid, M. (1989) vav, a novel human oncogene derived from a loxus ubiquitously expressed in hematopoietic cells. EMBO J. 8:2283-2290
21. Gulbins, E., Coggeshall, K.M., Baier, G., Katzav, S., Burn, P., and Altman, A. (1993) Tyrosine kinase-stimulated guanine nucleotide exchange activity of vav in T cell activation. Science 260:822-825
22. Gulbins, E., Langlet, C., Baier, G., Bonnefoy-Berard, N., Herbert, E., Altman, A., and Coggeshall, K.M. (1994) Tyrosine phosphorylation and activation of Vav GTP/GDP exchange activity in antigen receptor-triggered B cells. J. Immunol. 152:2123-2129
23. Wolf, M.L., Buckley, J.A., Goldfarb, A., Law, C.-L., and LeBien, T.W. (1991) Development of a bone marrow culture for maintence and growth of normal B cell precursors. J. Immunol. 147:3324-3330
24. Wormann, B., Anderson, J.M., Liberty, J.A., Gajl-Peczalska, K., Brunning, R.D., Silberman, T.L., Arthur, D.C., and LeBien, T.W. (1989) Establishment of a leukemic cell model for studying human pre-B to B cell differentiation. J. Immunol. 142:110-117
25. Goding, J.W. (1986) Monoclonal Antibodies: Principles and Practice, 2nd Ed., Academic Press, San Diego, CA
26. Lassoued, K., Nunez, C.A., Billips, L., Kubagawa, H., Monteiro, R.C., LeBien, T.W., and Cooper, M.D. (1993) Expression of surrogate light chain receptors is restricted to a late stage in pre-B cell differentiation. Cell 73:73-86
27. Ravichandran, K.S., Lee, K.K., Songyang, Z., Cantley, L.C., Burn, P., and Burakoff, S.J. (1993) Interaction of Shc with the
z chain of the T cell receptor upon T cell activation. Science 262:902-90528. Charest, D.L., Mordret, G., Harder, K.W., Jirik, F., and Pelech, S.L. (1993) Molecular cloning, expression, and characterization of the human mitogen-activated protein kinase p44
erk1. Mol. Cell. Biol. 13:4679-469029. Law, C.L., Sidorenko, S.P., Chandran, K.A., Draves, K.E., Chan, A.C., Weiss, A., Edelhoff, S., Disteche, C.M., and Clark, E.A. (1994) J. Biol. Chem. 269:12310-12319
30. Karnitz, L.M., Sutor, S.L., and Abraham, R.T. (1994) The Src-family kinase, Fyn, regulates the activation of phosphatidylinositol 3-kinase in an interleukin 2-responsive T cell line. J. Exp. Med. 179:1799-1808
31. Ren, C.L., Morio, T., Fu, S.M., and Geha, R.S. (1994) Signal transduction via CD40 involves activation of lyn kinase and phosphatidylinositol-3-kinase, and phosphorylation of phospholipase C
g2. J. Exp. Med. 179:673-68032. Bustelo, X.R., Ledbetter, J.A., and Barbacid, M. (1992) Product of vav proto-oncogene defines a new class of tyrosine protein kinase substrates. Nature 356:68-71
33. Leevers, S. J., and Marshall, C.J. (1992) Activation of extracellular signal-regulated kinase, ERK2, by p21
ras oncoprotein. EMBO J. 11:569-57434. de Vries-Smits, A.M.M., Burgering, B.M.Th., Leevers, S.J., Marshall, C.J., and Bos, J.L. (1992) Involvement of p21ras in activation of extracellular signal-regulated kinase 2. Nature 357:602-604
35. Alessandrini, A., Crews, C.M., and Erikson, R.L. (1992) Phorbol ester stimulates a protein-tyrosine/threonine kinase that phosphorylates and activates the Erk-1 gene product. Proc. Natl. Acad. Sci. USA 89:8200-8204
36. Uckun, F.M., and Ledbetter, J.A. (1988) Immunobiological differences between normal and leukemic human B-cell precursors. Proc. Natl. Acad. Sci. USA 85:8603-8607
37. Callard, R.E., Rigley, K.P., Smith, S.H., Thurstan, S., and Shields, J.G. (1992) CD19 regulation of human B cell responses. B cell proliferation and antibody secretion are inhibited or enhanced by ligation of the CD19 surface glycoprotein depending on the stimulating signal used. J. Immunol. 148:2983-2987
38. Uckun, F.M., Burkhardt, A.L., Jarvis, L., Jun, X., Stealey, B., Dibirdik, I., Myers, D.E., Turl-Ahlgren, L., and Bolen, J.B. (1993) Signal transduction through the CD19 receptor during discrete developmental stages of human B-cell ontogeny. J. Biol. Chem. 268:21272-21184
39. Chalupny, N.J., Kanner, S.B., Schieven, G.L., Wee, S., Gilliland, L.K., Aruffo, A., and Ledbetter, J.A. (1993) Tyrosine phosphorylation of CD19 in pre-B and mature B cells. EMBO J. 12:2691-2696
40. Coppola, J., Bryant, S., Koda, T., Conway, D., and Barbacid, M. (1991) Mechanism of activation of the vav protooncogene. Cell Growth Differ. 2:95-105
41. Katzav, S., Cleveland, J.L., Heslop, H.E., and Pulido, D. (1991) Loss of the amino-terminal helix-loop-helix domain of the vav proto-oncogene activates its transforming potential. Mol. Cell. Biol. 11:1912-1920
42. Katazv, S., Martin-Zanca, D., and Barbacid, M. (1989) vav, a novel human oncogene derived from a loxus ubiquitously expressed in hematopoietic cells. EMBO J. 8:2283-2290
43. Pleiman, C.M., Hertz, W.M., and Cambier, J.C. (1994) Activation of phosphatidylinositol-3' kinase by src-family kinase SH3 binding to the p85 subunit. Science 263:1609-1612
44. van Noesel, C.J.M., Lankester, A.C., van Schijndel, G.M.W., and van Lier, R.A.W. (1993) The CR2/CD19 complex on human B cells contains the src-family kinase Lyn. Int. Immunol. 5:699-705
45. Stokoe, D., Macdonald, S.G., Cadwallader, K., Symons, M., and Hancock, J.F. (1994) Activation of Raf as a result of recruitment to the plasma membrane. Science 264:1463-1467
46. Leevers, S.J., Paterson, H.F., and Marshall, C.J. (1994) Requirement for Ras in Raf activation is overcome by targeting Raf to the plasma membrane. Nature 369:411-414
47. Ray, L.B., and Sturgill, T.W. (1988) Insulin-stimulated microtubule-associated protein kinase is phosphorylated on tyrosine and threonine in vivo. Proc. Natl. Acad. Sci. USA 85:3753-3757
48. Lazarus, A.H., Kawauchi, K., Rapoport, M.J., and Delovitch, T.L. (1993) J. Exp. Med. 178:1765-1769
ACKNOWLEDGMENTS
We thank Dr. Julia G. Johnson for her technical assistance and valuable discussions; Dr. Frederick T. Boyd for his help with the densitometric quantitation; and Nisha Shah and Julie Rehmann Pribyl for technical assistance. We also thank Dr. Robert Abraham, Mayo Clinic, for his very helpful comments, and Dr. Che-Leung Law, University of Washington School of Medicine, for his critical review of the manuscript.
FOOTNOTES
* This work was supported by Grants R01 CA-31685 from the National Cancer Institute/National Institutes of Health and the Leukemia Task Force. W.-K. Weng is the recipient of a predoctoral fellowship from NIH Immunology Training Grant T32 AI-07313.
‡ Address correspondence and reprint requests to Tucker W. LeBien, PhD, Box 609 UMHC, Department of Laboratory Medicine and Pathology, University of Minnesota Medical School, 420 Delaware St. SE, Minneapolis, MN 55455
1 Abbreviations used in this paper: Ig, immunoglobulin; PTK, protein tyrosine kinase; PI 3-kinase, phosphatidylinositol 3-kinase; anti-p-Tyr, anti-phosphotyrosine mAb; MAP kinase, mitogen-activated protein kinase; MEK, mitogen-activated protein kinase and extracellular signal-regulated kinase kinase; mAb, monoclonal antibody; MHC, major histocompatability complex; PAGE, polyacrylamide gel electrophoresis; PMA, phorbol 12-myristate 13-acetate.